To assess the antihypertensive efficacy and safety of a fixed-dose combination of amlodipine and lisinopril (Amlopres-L) in Indian patients.
Postmarketing Surveillance Study on Amlopres-L (Amlodipine/Lisinopril combination)
19 Apr, 14
Aim
Study Patients
Patients with moderate hypertension or hypertension not controlled by amlodipine and atenolol alone (N=340).
Study Medication
Amlodipine 5 mg and lisinopril 5 mg once daily (Amlopres L)
Study Duration
4 weeks
Efficacy Parameters
- Responder rate defined as patients who achieved DBP ≤90 mmHg at the end of the study.
Results
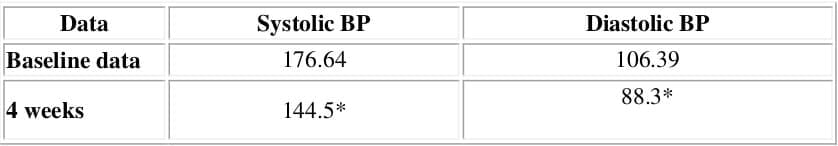
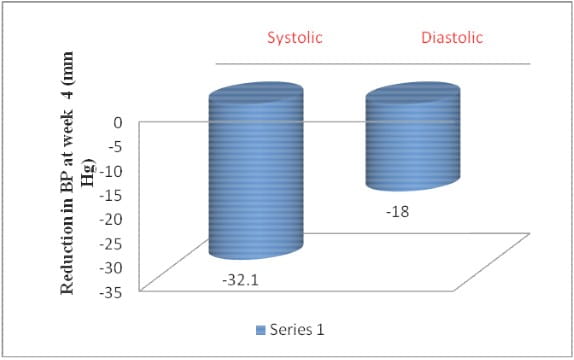
- Treatment with Amlopres-L showed significant reduction in blood pressure at end of 4 weeks
- At the end of therapy 78% of patients were classified as 'responders' to Amlopres-L therapy
- According to the study investigators Amlopres-L was 'very effective' in 69.7% patients, 'moderately effective' in 27.58% and poorly effective' in 2.7% patients
Safety
- Amlopres-L was safe in Indian patients with hypertension
- Treatment with Amlopres-L exhibited an excellent tolerability profile
- Commonly seen side effects were oedema, fatigue and headache
Conclusion
The once daily regimen of the fixed dose combination of amlodipine and lisinopril is safe in Indian patients with hypertension and results in clinically valuable additive antihypertensive effects.
The Indian Practitioner 1998: 51; 441-447