Treatment with Macitentan Reduced the Risk and Rate of Hospitalizations in Patients with PAH
Introduction
The Study with Endothelin Receptor Antagonist in Pulmonary arterial Hypertension (PAH) to Improve Clinical Outcome (SERAPHIN) trial demonstrated that macitentan, a dual endothelin-receptor antagonist, significantly reduced the mortality and morbidity in the patients with PAH. A composite secondary endpoint of “death due to PAH or hospitalization for PAH” was evaluated. The risk of such events was significantly reduced with macitentan. The results of the trial indicated a considerable impact on the PAH-related hospitalizations.
Aim
This study evaluated the effect of macitentan on the risk, rate, number of hospital days for all-cause and PAH-related hospitalizations. Additionally the risk and causes of hospitalizations unrelated to PAH were also assessed.
Methods
Study design
- Multicenter, double-blind, randomized, placebo-controlled, event-driven phase 3 trial
- Patients aged 12 years or more, with confirmed PAH and with 6-minute walk distance (6-MWD) of 50 m or more and class II, III or IV according to WHO functional classification were selected
- Patients could be treatment na?ve or receiving phosphodiesterase type 5 inhibitors, oral or inhaled prostanoids, calcium channel blockers or L-arginine
- The cohort was randomized into 3 groups after screening to receive placebo once daily, macitentan 3 mg once daily or macitentan 10 mg once daily
Endpoints
- Time to first all-cause hospitalization
- Time to first PAH-related hospitalization upto the end of treatment
- Total number of hospital days per patient-year for both all-cause and PAH-related hospitalizations
- Time to first non-PAH-related hospitalization and their causes
Results
- The overall cohort comprised of 742 patients
- 250 received placebo, 250 received macitentan 3 mg and 242 received macitentan 10 mg
- The cohort was characterized by 76.5% females, mean age of 45.6+16.1 years, 55% had idiopathic PAH
- Mean time from PAH diagnosis was 2.7+4.0 years
- Overall median duration of treatment was 115 weeks
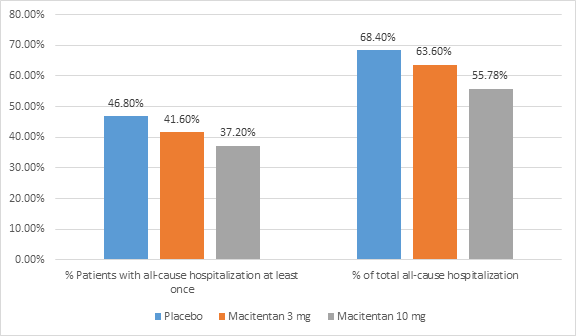
- The rate of all-cause hospitalization at least once during the treatment and % of total all-cause hospitalizations is shown in figure 1
- The risk of all-cause hospitalization with 3 mg macitentan was reduced by 18.9% (HR=0.811; p=0.1208) and by 32.3% with macitentan 10 mg (HR=0.677; p=0.0051) as compared to placebo
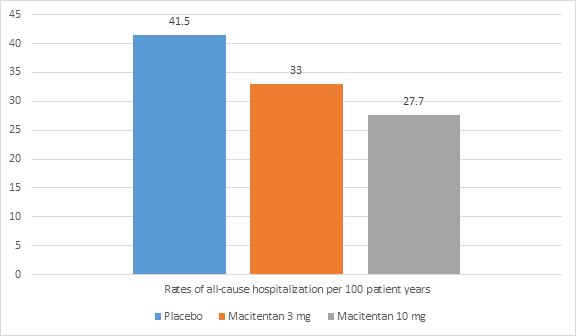
- Rates of all-cause hospitalization per 100 patient years across all the groups is shown in figure 2.
- There was a reduction in the macitentan groups as compared to placebo with respect to
- rate of all-cause hospitalization
- number of hospital days
- risk of PAH-related hospitalization
- rate of PAH-related hospitalization
- number of hospital days due to PAH
|
Outcome |
Macitentan 3 mg group |
Macitentan 10 mg group | ||
|
|
% Reduction |
p value |
% Reduction |
p value |
|
All-cause hospitalization |
20.5% |
0.0378 |
33.1% |
0.0005 |
|
No of hospital days |
30.6% |
0.0278 |
31.0% |
0.0336 |
|
Risk of PAH-related hospitalization |
42.7% |
0.0015 |
51.6% |
<0.0001 |
|
Rate of PAH-related hospitalization |
44.5% |
0.0004 |
49.8% |
<0.0001 |
|
No of hospital days due to PAH |
53.3% |
0.0001 |
52.3% |
0.0003 |
- There were no significant differences in the risk of non-PAH-related hospitalization between the groups
Conclusion
- Treatment with macitentan 10 mg resulted in significant reduction in the risk and rate of all-cause hospitalization in patients with pulmonary arterial hypertension.
- Macitentan 10 mg also significantly reduced the duration of hospital stay
- The risk and rate of PAH-related hospitalization was also reduced significantly in the patients treated with macitentan
J Am Coll Cardiol HF. 2015;3:1-8.